Work Function and the electron escape energy
Work Function is often confused with escape energy of an electron.
Those two concepts are related but not the same.
Escape energy
The escape energy of an electron from bulk of a conductor through a given surface is the minimum energy that needs to be delivered to an electron near the Fermi level of the conductor to move it outside of the material.
This energy is affected by the surface potentials created by:
- surface dipole layers and/or by
- surface unbalanced charge densities
because both of those quantities do shift the Fermi level of the conductor with respect to the lowest vacuum level. One can see that the escape energy is a function of both: the properties of the conductor’s material and the conditions (extra charges) it is currently in. In particular, if the conductor is positively charged, the escape energy increases because the escaping electron must overcome the pull of the positive charge.
The Work Function
The Work Function, on the other hand, is a property of the conducting material alone. Namely, the Work Function is equal to the electron escape energy through a surface, only if the excess charge density of that surface is zero.
This distinction between Work Function and the escape energy has profound consequences for measurement techniques that can be employed to determine the Work Function of a material.
In order to perform such a measurement of a Work Functions through a surface, one needs to make sure that that surface is not charged. In order to remove excess charge from such a surface an appropriate electric voltage $V^0$ needs to be applied between the material and a ground. The voltage shifts the Fermi level of the material with respect to the vacuum energy levels. See the Electrochemical vs chemical potential post. Thus, the Work Function through a given surface can be defined as the distance in energy between the material’s Fermi level and the bottom of vacuum band
$$
\Phi = E_{\mbox{vac}} - E_F
$$ provided that the surface has no excess charge i.e., a proper potential is applied to the conductor. This way one maintains different Work Functions through different surfaces of the same body. It is possible because the Fermi level $E_F$ is a function of the voltage $V^0$ applied to remove the excess charge from the surface of interest
$$
\Phi = E_{\mbox{vac}} - E_F\left( V^0 \right ).
$$ If one assumes the $E_{\mbox{vac}}=0$ convention then the last expression simplifies into
$$
\Phi = -E_F\left (V^0\right )
$$ and the Work Function is given by just the Fermi level.
This simple expression captures all the nuanced aspects of the Work Functions concept. It includes effects related to a double layers that might be present at the surface or adsorbates modifying the surface charge distribution.
The double layer itself has zero electric charge but it generates an electric potential that affects the surface charge distribution on conductors.
IUPAC Recommendations 1985
The zero charge density approach to the Work Function is honored in the old IUPAC “electron work function” definition

It was put together by Sergio Trasatti and is a rare example of deeper, although still incomplete, understanding of the subject.
Incorrect definitions of Work Function
C. Kittel Introduction to Solid State Physics
In the 8th edition of Charles Kittel’s famous Introduction to Solid State Physics, page 494 introduces the Work Function as
the difference in potential energy of an electron between the vacuum level and
the Fermi level. The vacuum level is the energy of an electron at rest at a point
sufficiently far outside the surface so that the electrostatic image force on the
electron may be neglected-more than 100 Å from the surface. The Fermi
level is the electrochemical potential of the electrons in the metal.
This approach admits that the Fermi level, defined as electrochemical potential, is sensitive to overall excess charge of a material. Thus it confuses the electron escape energy with the Work Function.
Wikipedia (as of March 31st, 2025)
Wikipedia’s definition follows that of Kittel’s from the section above with one caveat: now the surface charge density is implicitly removed with the $-e\phi$ term

However the term $-e\phi$ IS certainly NOT the “energy of an electron at rest in the vacuum nearby the surface”!
See the discussion below.
New IUPAC Recommendations 2021
Regrettably, IUPAC has regressed from the approach in Recommendations 1985 still available as “electron work function” to the definition cited below under the term “work function”
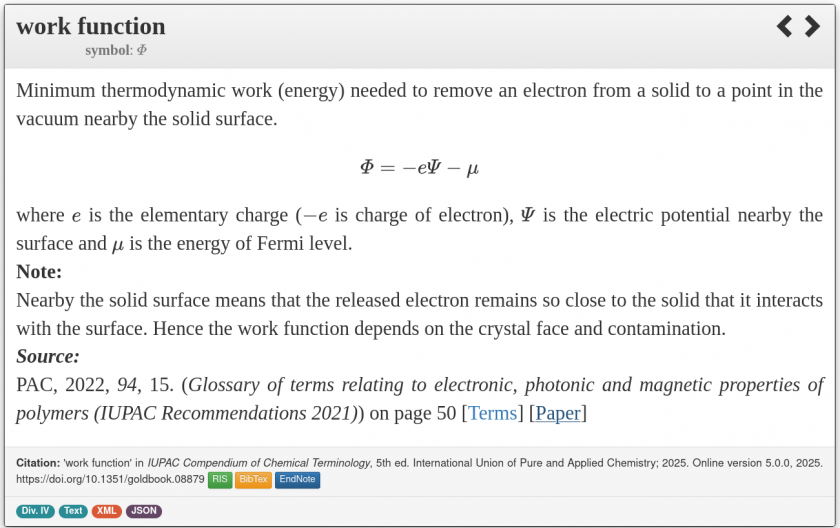
Here several other issues are introduced.
The unfortunate phrasing of this definition, in the very first sentence, states that the electron removed from the material is still close to the surface. That would mean that the attractive image charge force of the material has not been overcome yet and there is no equilibrium reached! So, Work Function would be a non-equilibrium quantity all of a sudden. It its absurd.
But suppose that the electron for the Work Function account needs to be removed to infinity after all and the “nearby the surface” only pertains to the definition of the potential $\Psi$. The $\Psi$ would be, so called, Volta potential that is important in the definition of Contact Potential Difference.
Then the vibe physics explanation is delivered in the “Note”. According to the Note the $\Psi$ must be electric potential experienced by an electron “nearby the solid surface”. Authors are undeterred by the fact that any electric potential in physics is derived from electric field. And the electric field is the force experienced by a test charge in presence of other charged bodies. The test charge is not an electron! It is an infinitesimally small electric charge that does not disturb other (possibly mobile) charges in the system. An electron “nearby the solid surface” of a conductor will rearrange the surface carriers density of the material (the image charge effect), so the concept of electric potential is lost.
The image charge effect cannot be neglected because, together with the surface dipole, it determines the Work Function of any material.